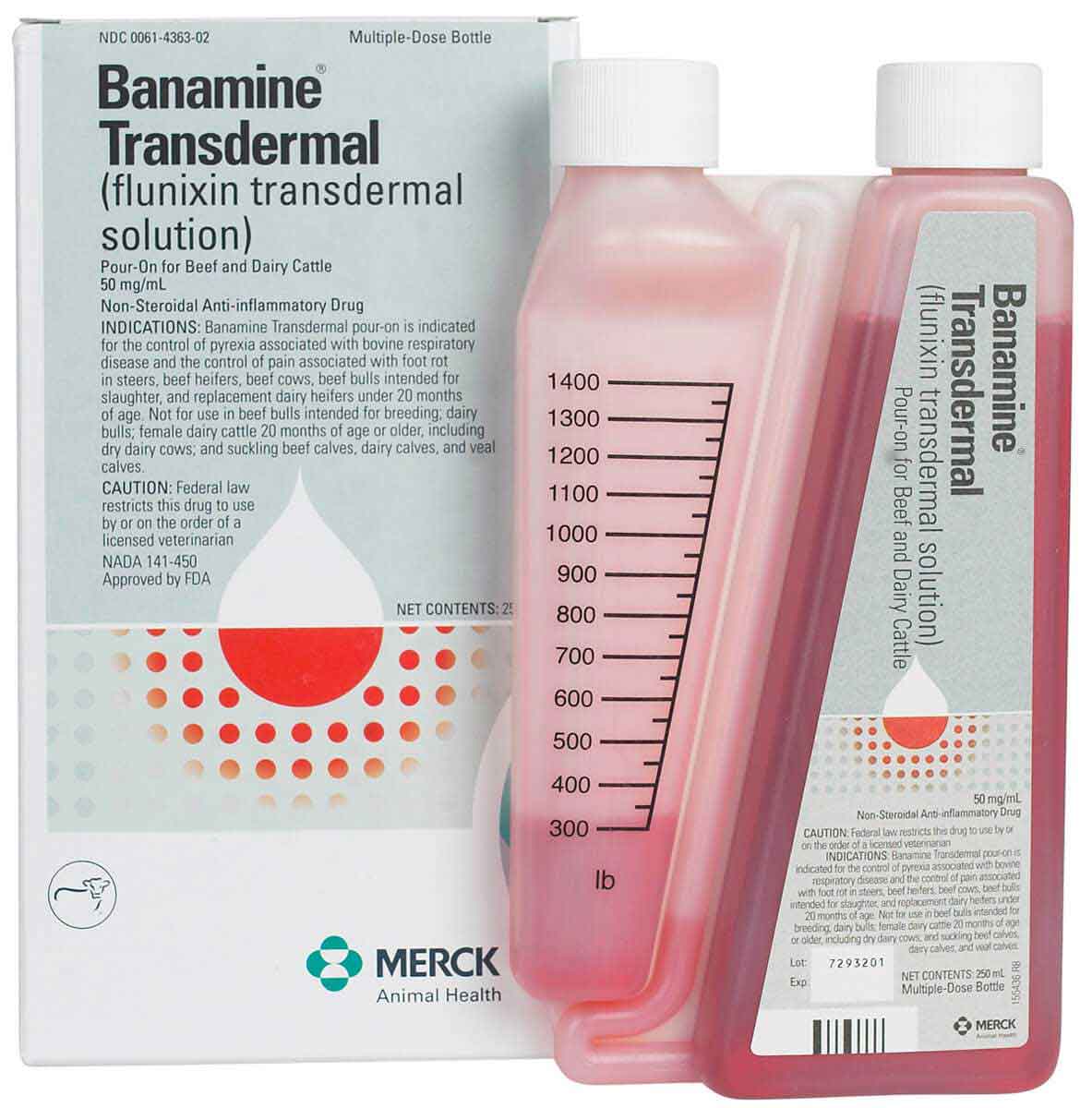
Description
Banamine Transdermal pour-on is indicated for the control of pyrexia associated with bovine respiratory disease and acute bovine mastitis, and the control of pain associated with foot rot in beef cattle 2 months of age and older and dairy cattle.
CAUTION:
Federal law restricts this drug to use by or on the order of a licensed veterinarian.
DESCRIPTION: Each milliliter of Banamine Transdermal pour-on contains 50 mg flunixin (equivalent to 83 mg flunixin meglumine), 150 mg pyrrolidine, 50 mg L-menthol, 500 mg propylene glycol dicaprylate/dicaprate NF, 0.20 mg FD&C Red No. 40, and glycerol monocaprylate NF qs.
Not for use in beef and dairy bulls intended for breeding over 1 year of age, replacement dairy heifers over 20 months of age, dry dairy cows, dairy calves, or veal calves.
DOSAGE AND ADMINISTRATION:
Apply only once at a dose of 3.3 mg flunixin per kg body weight (1.5 mg/lb; 3 mL per 100 lbs) topically in a narrow strip along the dorsal midline from the withers to the trailhead. Round the dose up to the nearest weight increment on the dosing chamber. If pyrexia, or the pain associated with foot rot persists, the diagnosis should be re-evaluated and alternative therapy considered. Do not treat cattle if the hide is wet or may get wet in the six hours after dosing because effectiveness has not been evaluated under wet hide conditions.
Practice the Administration and Overfill Reduction Instructions a few times to become familiar with operating the package before dosing animals.
If the dosing chamber is overfilled follow the Overfill Reduction Instructions.
A small amount of liquid will remain on the walls of the chamber, but the chamber is calibrated to account for this.
OVERFILL REDUCTION INSTRUCTIONS
Tilt the bottle to allow an air pocket to form at the beginning of the transfer tube inside the bottle.
Hold the bottle horizontally to allow product to cover the end of the transfer tube inside the dosing chamber.
Figure 1 – Recommended pour-on location
CONTRAINDICATIONS: NSAIDs inhibit production of prostaglandins which are important in signaling the initiation of parturition. The use of flunixin can delay parturition and prolong labor which may increase the risk of stillbirth. Do not use Banamine Transdermal pour-on within 48 hours of expected parturition. Do not use in animals showing hypersensitivity to flunixin meglumine.
USER SAFETY WARNINGS: Not for use in humans. Keep out of reach of children. Flunixin transdermal solution is a potent non-steroidal anti-inflammatory drug (NSAID), and ingestion may cause gastrointestinal irritation and bleeding, kidney, and central nervous system effects.
This product has been shown to cause severe and potentially irreversible eye damage (conjunctivitis, iritis, and corneal opacity) and irritation to skin in laboratory animals. Users should wear suitable eye protection (face shields, safety glasses, or goggles) to prevent eye contact; and chemical resistant gloves and appropriate clothing (such as long-sleeve shirt and pants) to prevent skin contact and/or drug absorption. Wash hands after use.
In case of accidental eye contact, flush eyes immediately with water and seek medical attention. If wearing contact lenses, flush eyes immediately with water before removing lenses. In case of accidental skin contact and/or clothing contamination, wash skin thoroughly with soap and water and launder clothing with detergent. In case of ingestion do not induce vomiting and seek medical attention immediately. Probable mucosal damage may contraindicate the use of gastric lavage. Provide product label and/or package insert to medical personnel.
|
|
Withdrawal Periods and Residue Warnings: Milk that has been taken during treatment and for 48 hours after treatment must not be used for human consumption. Cattle must not be slaughtered for human consumption within 8 days of treatment. Not for use in replacement dairy heifers 20 months of age or older or dry dairy cows; use in these cattle may cause drug residues in calves born to these cows or heifers. Not for use in beef calves less than 2 months of age, dairy calves, and veal calves. A withdrawal period has not been established for this product in pre-ruminating calves. Approved only as a single topical dose in cattle. Repeated treatments may result in violative residues in milk or in edible tissues. |
|
PRECAUTIONS: As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal, renal, and hepatic toxicity. Sensitivity to drug-associated adverse events varies with the individual patient. Patients at greatest risk for adverse events are those that are dehydrated, on concomitant diuretic therapy, or those with renal, cardiovascular, and/or hepatic dysfunction. Banamine Transdermal should be used with caution in animals with suspected pre-existing gastric erosions or ulcerations. Concurrent administration of other NSAIDs, corticosteroids, or potentially nephrotoxic drugs should be avoided or used only with careful monitoring because of the potential increase of adverse events. Banamine Transdermal is approved only as a single topical dose. The safety of repeated treatment has not been evaluated.
NSAIDs are known to have potential effects on both parturition (see Contraindications) and the estrous cycle. There may be a delay in the onset of estrus if flunixin is administered during the prostaglandin phase of the estrous cycle. NSAIDs are known to have the potential to delay parturition through a tocolytic effect. The use of NSAIDs in the immediate post-partum period may interfere with uterine involution and expulsion of fetal membranes. Cows should be monitored carefully for placental retention and metritis if Banamine Transdermal pour-on is used within 24 hours after parturition.
Not for use in beef and dairy bulls intended for breeding over 1 year of age because reproductive safety has not been evaluated. BMK OIL





Reviews
There are no reviews yet.